A Clinical Overview for Practice
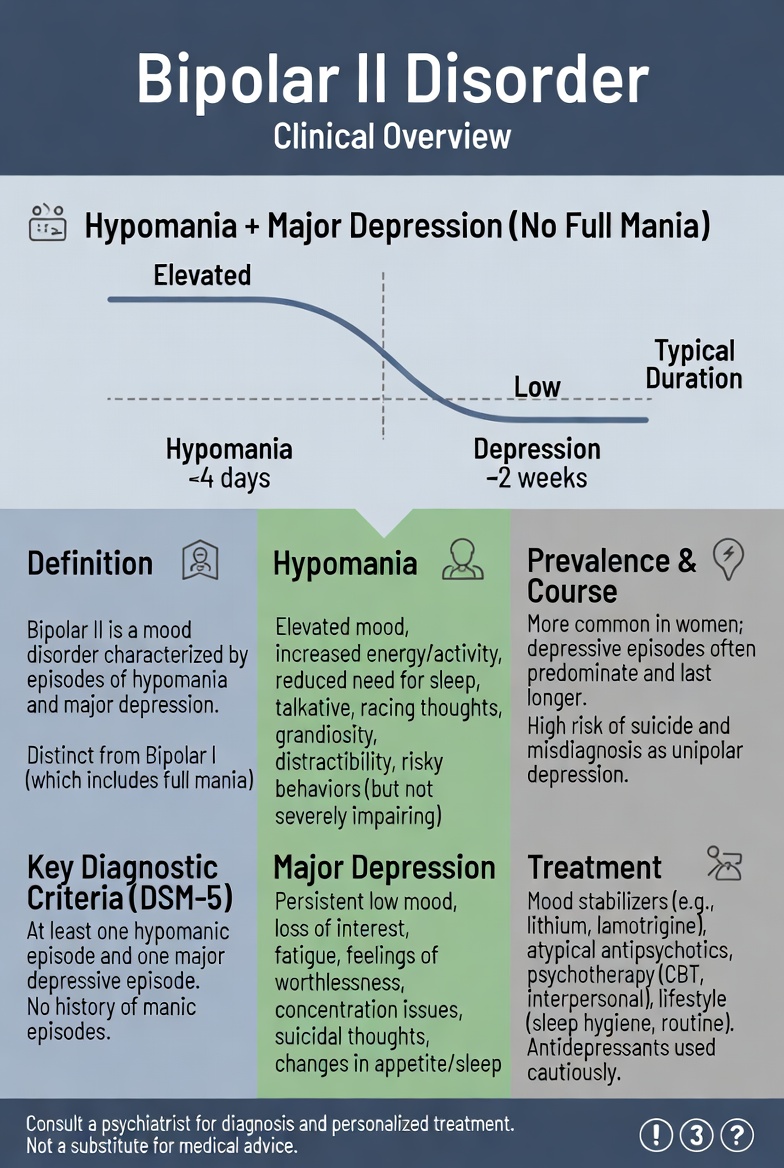
Bipolar II Disorder (BD-II) is a chronic, episodic mood disorder characterized by at least one hypomanic episode and at least one major depressive episode, with no lifetime history of full mania (APA, 2022). Despite being clinically distinct from Bipolar I Disorder (BD-I), BD-II is persistently mischaracterized as a milder or sub-threshold condition — an assumption that current evidence firmly refutes.
BD-II carries substantial functional impairment, a high burden of cognitive dysfunction, and an elevated suicide risk that is statistically equivalent to BD-I (Dev et al., 2024). The longitudinal course is predominantly depressive: depressive episodes outnumber hypomanic episodes by a ratio of approximately 39:1 (Berk et al., 2025). The disorder is also substantially underdiagnosed, with average diagnostic delays of five to ten years, most commonly due to initial misclassification as major depressive disorder (MDD) (Berk et al., 2025).
This misdiagnosis has direct clinical consequences. Antidepressant monotherapy — the most common treatment error following an MDD misdiagnosis — can worsen outcomes by accelerating cycling and destabilizing mood. The Indian Journal of Psychiatry Clinical Practice Guidelines emphasize that BD-II remains persistently undertreated and underrecognized, underscoring the importance of clinician awareness, thorough longitudinal history-taking, and validated screening tools to close the diagnostic gap (Shah et al., 2017).
This clinical overview covers the assessment, diagnosis, treatment planning, implementation, and evaluation of BD-II, with the goal of improving understanding and practice across disciplines.
Epidemiology
Prevalence
Bipolar disorder as a spectrum affects an estimated 40 million people worldwide (Hu et al., 2025). The lifetime prevalence of BD-II specifically is estimated at 0.4 to 1.1% globally. While BD-II is less common than BD-I in clinical samples, it is actually more prevalent in community surveys — largely because hypomanic episodes frequently go unreported and undetected (Berk et al., 2025).
In the United States, the combined lifetime prevalence of BD-I and BD-II is estimated at 4.4%, with BD-II being the more prevalent subtype in community-based studies (Hu et al., 2025). According to Global Burden of Disease (GBD) 2021 data, global bipolar disorder incidence rose from approximately 30.2 million cases in 1990 to 53.9 million in 2021, with projections of continued increase through 2035 (Hu et al., 2025). BD-II contributes substantially to this burden through high rates of depressive disability and the extensive years lived with disability (YLDs) it generates.
Patterns
BD-II typically begins in late adolescence to early adulthood, with a mean age of onset of approximately 20 to 22 years. Onset may occur later, particularly in women presenting with a depression-first pattern (Berk et al., 2025). Unlike BD-I, which affects men and women in equal proportions, BD-II shows a female predominance. Women are more likely to experience rapid cycling, a depression-polarity predominance, and postpartum presentations (Berk et al., 2025).
The longitudinal course is characterized by depressive dominance: patients spend approximately three times as many weeks in depressive states as in hypomanic states (Berk et al., 2025). Seasonal patterns occur in some individuals, with depressive episodes more prevalent during autumn and winter. Diagnostic delay is a consistent epidemiological feature across all global regions, with average delays of five to ten years reported internationally (Berk et al., 2025).
Determinants
Etiology
BD-II arises from complex gene-environment interactions. Its pathophysiology overlaps substantially with BD-I but includes distinct features, such as lower genome-wide association study (GWAS)-identified genetic loadings and stronger links to depressive temperament and cyclothymic traits (Berk et al., 2025; Mullins et al., 2021).
Biological Factors
- Heritability is estimated at 60 to 80% for the bipolar spectrum. A GWAS of more than 40,000 bipolar cases identified 64 genomic risk loci, with overlap between BD-I and BD-II but also subtype-specific genetic variants (Mullins et al., 2021).
- Pathophysiological mechanisms under active investigation include neuroinflammation, circadian rhythm dysregulation, and mitochondrial dysfunction. These mechanisms are shared with BD-I but may manifest differently in BD-II (Berk et al., 2025).
- Neuroimaging studies document frontolimbic connectivity disruption and amygdala volume changes, though BD-II-specific neuroimaging findings are less well-established than those for BD-I (Berk et al., 2025).
- Circadian clock dysregulation is a core biological vulnerability in BD-II. Disruptions in sleep-wake cycles precipitate both hypomanic and depressive episodes, making circadian rhythm stabilization a key target for pharmacotherapy and Interpersonal and Social Rhythm Therapy (IPSRT) (Berk et al., 2025).
Psychological Factors
- Reward hypersensitivity and behavioural activation system (BAS) dysregulation are implicated in hypomanic vulnerability.
- Childhood trauma, adverse life events, and insecure attachment patterns are associated with earlier onset, longer depressive episodes, and more frequent cycling.
- Cyclothymic temperament is a strong premorbid predictor of BD-II, occurring in approximately 50% of cases (Berk et al., 2025).
- Poor illness insight and alexithymia may contribute to underreporting of hypomanic symptoms and to diagnostic delay.
Social Factors
- Social rhythm disruption — including irregular sleep-wake cycles, shift work, and stressful interpersonal events — precipitates mood episodes. Addressing social rhythms is a core target of IPSRT.
- Low socioeconomic status, social isolation, and limited access to community mental health resources are associated with diagnostic delay and worse outcomes.
- Emerging evidence links exposure to air pollution and climate change to worsening bipolar outcomes (Radua et al., 2024).
Risk Factors
- Family history of bipolar disorder or recurrent depression — the strongest predictor for early identification.
- Cyclothymic or hyperthymic temperament during adolescence.
- Substance use disorders, which co-occur in approximately 30.7% of patients with bipolar disorder (Léda-Rêgo et al., 2024).
- History of multiple antidepressant trials with incomplete response or treatment-emergent activation.
- Postpartum onset — BD-II is a significant contributor to postpartum psychiatric illness.
- Female sex, given the depression-predominant phenotype that is common in BD-II.
Effects on Population
Individual
BD-II is associated with significant and often underestimated functional impairment. Cognitive deficits encompassing attention, memory, and executive function persist even during euthymia and contribute to occupational disability (Berk et al., 2025). Suicide risk in BD-II is high and equivalent to that in BD-I. A 2024 systematic review and meta-analysis found a pooled odds ratio of 1.00 (95% CI: 0.75 to 1.34) for completed suicide in BD-II compared to BD-I, directly dispelling the clinical assumption that BD-II carries lesser suicide risk (Dev et al., 2024). Lifetime suicide attempt prevalence in BD-II is approximately 32% (Berk et al., 2025).
Misdiagnosis as MDD, followed by antidepressant monotherapy, is associated with increased cycling frequency, the emergence of mixed features, and a higher risk of suicidal behavior (Berk et al., 2025; Shah et al., 2017).
Family
Families of individuals with BD-II bear a significant caregiver burden, even though the condition has lower clinical visibility than BD-I The predominantly depressive course means caregivers commonly witness prolonged withdrawal, functional decline, and suicidality — experiences that are emotionally and practically demanding. Children of a parent with BD-II carry elevated risk for mood and anxiety disorders. Family psychoeducation and family-focused therapy are evidence-based approaches for addressing these impacts (Levrat et al., 2024; Miklowitz et al., 2021).
Community
BD-II contributes substantially to community-level psychiatric morbidity through high rates of depression-related disability, frequent emergency department presentations, and costly cycles of misdiagnosis and inappropriate treatment. Psychiatric comorbidities are common: lifetime prevalence of anxiety disorders is approximately 40.4%, substance use disorders 30.7%, and ADHD 18.6% in patients with bipolar disorder (Léda-Rêgo et al., 2024). Stigma and misconceptions about BD-II as a mild or personality-based condition perpetuate delayed help-seeking and suboptimal resource allocation (Berk et al., 2025; Shah et al., 2017).
Assessment
Symptom Severity Assessment Tools
Preclinical / Screening
The Mood Disorder Questionnaire (MDQ) is the most widely validated and recommended self-report screening tool for bipolar spectrum disorders, including BD-II. It is recommended prior to initiating antidepressants for what appears to be unipolar depression (Keramatian et al., 2023; Shah et al., 2017). The Hypomania Checklist-32 (HCL-32) has demonstrated superior sensitivity for BD-II compared to the MDQ because of its focus on hypomanic rather than manic features; it is an appropriate complementary screen in suspected BD-II cases (Berk et al., 2025). The Patient Health Questionnaire-9 (PHQ-9) is suitable for monitoring depressive symptom severity but must not be used as a standalone BD-II screen because it does not capture hypomanic features.
A key clinical caveat: patients with BD-II frequently present during a depressive phase and do not spontaneously report prior hypomanic episodes. Collateral history from a reliable informant is therefore critical at every assessment (Shah et al., 2017).
Clinical — Mental Status Examination (MSE)
A thorough MSE is essential at each clinical encounter, with specific attention to the subtleties of hypomania — symptoms that patients often miss or normalize. Key domains to assess include the following.
- Appearance and Behaviour: Slightly increased energy, reduced inhibition, mildly decreased need for sleep during hypomania; psychomotor slowing, reduced grooming, or tearfulness during depression.
- Speech: Mildly pressured or more spontaneous than usual during hypomania; slowed, low-volume, with reduced spontaneity during depression.
- Mood and Affect: Elevated, bright, or mildly irritable during hypomania; dysphoric, anhedonic, or anxious during depression. Clinicians must distinguish reactive mood brightening from sustained hypomanic elevation.
- Thought Form: Slightly accelerated thought process during hypomania; slowed or ruminative during depression.
- Thought Content: Mildly inflated self-esteem, increased confidence, and future-oriented planning during hypomania; worthlessness, hopelessness, and passive death wishes or suicidal ideation during depression.
- Perceptions: Psychotic features are absent in hypomania by definition (APA, 2022). Their presence mandates reclassification to BD-I or another diagnosis.
- Cognition: Distractibility during hypomania; concentration difficulties and memory complaints during depression. Formal neurocognitive testing (MoCA, BACS) is indicated if euthymic cognitive impairment is suspected.
- Insight and Judgment: Insight is typically preserved or only mildly reduced during hypomania — a key distinguishing feature from BD-I mania.
Clinician-Rated Scales
The Hypomania Interview Guide (HIGH-C) is recommended for assessing hypomanic severity. The Montgomery-Asberg Depression Rating Scale (MADRS) or the Hamilton Rating Scale for Depression (HDRS-17) are preferred for depressive severity, given their sensitivity to BD-associated symptom change. The Young Mania Rating Scale (YMRS) is less sensitive to hypomania due to its mania-weighted item anchors.
Post-Clinical / Monitoring
The PHQ-9 is appropriate for regular monitoring of depressive episodes between clinical visits. Life chart methodology, structured, prospective mood tracking, is recommended to identify longitudinal patterns, episode triggers, and treatment response. This tool is particularly valuable in BD-II given its predominantly depressive course (Keramatian et al., 2023). Digital mood-monitoring applications are an emerging adjunct that may improve detection of subtle hypomanic shifts (Levrat et al., 2024). Regular sleep diary monitoring is recommended given the central role of circadian rhythm disruption in BD-II (Berk et al., 2025).
Diagnosis of Bipolar II Disorder
DSM-5-TR Diagnostic Criteria for Bipolar II Disorder (APA, 2022)
BD-II requires the lifetime presence of at least one hypomanic episode (Criteria A through F) and at least one major depressive episode, with no history of a full manic episode. The presence of even one manic episode, regardless of context upgrades the diagnosis to BD-I.
Criterion A: Hypomanic Episode: Mood and Energy
A distinct period of abnormally and persistently elevated, expansive, or irritable mood AND abnormally and persistently increased goal-directed activity or energy, lasting at least four consecutive days and present most of the day, nearly every day.
Criterion B: Symptom Cluster (DIGFAST)
During the period of mood and energy disturbance, three or more of the following symptoms must be present (four or more if mood is only irritable), representing a noticeable change from usual behaviour:
- D — Distractibility: Attention easily drawn to unimportant or irrelevant stimuli.
- I — Impulsivity / increased goal-directed activity (social, occupational, academic, or sexual) OR psychomotor agitation.
- G — Grandiosity: Inflated self-esteem or grandiosity.
- F — Flight of ideas or subjective experience of racing thoughts.
- A — Activity increase / psychomotor agitation.
- S — Sleep decreased: Decreased need for sleep without associated fatigue (e.g., feeling rested after only three hours).
- T — Talkativeness: More talkative than usual, or experiencing pressure to keep talking.
Criterion C: Observable Change Without Severe Impairment
This is a critical distinction from BD-I. The hypomanic episode must be associated with an unequivocal change in functioning that is observable by others, but the episode must not be severe enough to cause marked impairment in social or occupational functioning, and must not require hospitalization. The complete absence of psychotic features is mandatory; their presence would indicate a manic episode and a BD-I diagnosis (APA, 2022).
Criterion D: Not Substance- or Medical Condition-Induced
The episode must not be attributable to the physiological effects of a substance or another medical condition. Importantly, if a full hypomanic episode emerges and persists beyond the expected physiological effect of antidepressant treatment, it counts toward a BD-II diagnosis.
Criterion E: Major Depressive Episode Required
Criteria must be met for at least one major depressive episode: five or more of the following symptoms must be present during the same two-week period and represent a change from previous functioning, with at least one symptom being either (1) depressed mood or (2) loss of interest or pleasure:
- Depressed mood most of the day, nearly every day.
- Markedly diminished interest or pleasure in all, or almost all, activities.
- Significant weight change or appetite disturbance.
- Insomnia or hypersomnia.
- Psychomotor agitation or retardation observable by others.
- Fatigue or loss of energy.
- Feelings of worthlessness or excessive or inappropriate guilt.
- Diminished ability to concentrate or make decisions.
- Recurrent thoughts of death, suicidal ideation, or a suicide attempt.
Criterion F: No History of Mania
There has never been a manic episode. This is the defining diagnostic boundary between BD-II and BD-I (APA, 2022).
Specifiers
Clinicians should apply all relevant specifiers. These include: with anxious distress; with mixed features (which, in BD-II, are associated with elevated suicide risk); with rapid cycling (four or more mood episodes per 12-month period); with peripartum onset; with seasonal pattern; and course specifiers indicating whether the patient is in partial remission, full remission, or experiencing a current hypomanic or depressive episode.
Differential Diagnosis
Bipolar I Disorder
BD-I is the critical differential. It requires at least one lifetime manic episode. If a patient’s reported ‘hypomanic’ episode caused marked functional impairment, required hospitalization, or was accompanied by psychotic features, the diagnosis must be BD-I. A thorough longitudinal history and collateral information are essential. A single missed manic episode completely alters both the diagnosis and treatment plan (APA, 2022).
Major Depressive Disorder (MDD)
MDD is the most common misdiagnosis for BD-II. Patients with BD-II frequently present for treatment during depressive phases and may not volunteer a hypomanic history, either through lack of illness insight or through normalizing their hypomanic states. Clinicians must proactively screen for past hypomanic episodes using validated tools such as the MDQ and HCL-32, as well as collateral history. Prescribing antidepressant monotherapy on the basis of an MDD diagnosis carries the risk of precipitating mixed features, accelerating cycling, and triggering mood switches (Berk et al., 2025; Shah et al., 2017).
Cyclothymic Disorder
Cyclothymic disorder involves numerous periods of hypomanic and depressive symptoms that do not meet full criteria for a hypomanic or major depressive episode, persisting for at least two years. When criteria for both a hypomanic episode and a major depressive episode are met at any point, BD-II takes diagnostic precedence (APA, 2022). Cyclothymic temperament is a significant premorbid predictor of BD-II (Berk et al., 2025).
Borderline Personality Disorder (BPD)
BPD is one of the most clinically challenging differentials for BD-II. Overlapping features include affective instability, impulsivity, interpersonal sensitivity, and suicidal behaviour. There are several key distinguishing features. In BPD, affective shifts are typically reactive to interpersonal stressors and last hours to days; in BD-II, mood episodes last days to weeks without requiring an interpersonal trigger. BD-II hypomanic episodes involve increased energy and decreased sleep need — features that are absent in BPD. It is also important to note that borderline personality traits are significantly more frequent and severe in BD-II than in BD-I, and both conditions commonly co-occur (Berk et al., 2025). Longitudinal assessment over multiple encounters is the most reliable differentiating strategy.
Attention-Deficit/Hyperactivity Disorder (ADHD)
ADHD is a chronic, non-episodic neurodevelopmental disorder with childhood onset. Distractibility, impulsivity, and emotional dysregulation overlap with BD-II hypomanic features. The key distinction is that ADHD lacks the episodic, time-limited nature of hypomania; elevated mood, decreased sleep need, grandiosity, and flight of ideas are not features of ADHD. Comorbid ADHD occurs in approximately 18.6% of patients with bipolar disorder (Léda-Rêgo et al., 2024) and requires independent assessment and treatment.
Substance-Induced Mood Disorder
Stimulant intoxication (cocaine, amphetamine, MDMA) and alcohol-related mood states can mimic hypomanic episodes. A BD-II diagnosis requires that the hypomanic episode persist beyond the expected physiological effect of the substance. Urine drug screening and a detailed substance use history are mandatory at initial assessment. Comorbid substance use disorders affect approximately 30.7% of patients with bipolar disorder (Léda-Rêgo et al., 2024).
Unipolar Depression with Antidepressant-Emergent Activation
Antidepressant-induced activation states is characterized by increased energy, irritability, reduced sleep, and risk-taking behaviour — may resemble hypomania. These states must be distinguished from a true hypomanic episode on the basis of persistence beyond the pharmacological effect of the medication and the patient’s temporal pattern. A prior history of spontaneous hypomanic episodes is the key differentiating factor (APA, 2022).
Plan for Bipolar II Disorder
Treatment Goals
Consistent with the 2023 Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) guidelines (Keramatian et al., 2023) and the Indian Journal of Psychiatry Clinical Practice Guidelines (Shah et al., 2017), the overarching goals of BD-II management are as follows.
- Acute stabilization: Achieve full remission of the current hypomanic or depressive episode and ensure patient safety. Suicide risk assessment must receive particular attention, given the high prevalence of suicidal ideation and attempts in BD-II.
- Continuation: Prevent relapse in the short-to-medium term, typically defined as four to six months following episode remission.
- Maintenance: Prevent future depressive and hypomanic recurrences. Untreated BD-II shows an average emotional relapse rate of approximately 0.44 episodes per year.
- Functional restoration: Address cognitive impairment, restore occupational and interpersonal functioning, and improve quality of life.
- Avoidance of iatrogenic harm: Antidepressant monotherapy is specifically contraindicated, as it may worsen BD-II by destabilizing mood and increasing cycling frequency (Berk et al., 2025; Shah et al., 2017).
Implementation of Treatment Plan for Bipolar II Disorder
Psychopharmacology
Pharmacotherapy for BD-II must be phase-specific and guided by episode polarity, comorbidities, and patient preference. Evidence ratings below are derived from the CANMAT/ISBD 2023 guidelines (Keramatian et al., 2023). It is important to note that BD-II-specific pharmacotherapy evidence is substantially less robust than for BD-I; many recommendations are extrapolated from BD-I trials or from heterogeneous bipolar samples.
First-Line Pharmacotherapy
Acute Hypomanic Episode
- Lithium (Level 2): Indicated for acute hypomania. The evidence base is lower in intensity than for mania, but lithium is effective and preferred given its maintenance benefits. Target serum level for BD-II maintenance is 0.6 to 0.8 mEq/L (Shah et al., 2017).
- Quetiapine (Level 2): Effective for both the depressive and hypomanic poles of BD-II. Particularly useful when rapid symptom control is needed.
- Valproate (Level 3): May be used for hypomanic episodes, especially when agitation or mixed features are present. Must be avoided in women of reproductive age due to significant teratogenicity and neurodevelopmental risks.
- Behavioural strategies: Regularizing sleep-wake schedules and reducing stimulation are first-line non-pharmacological measures for mild-to-moderate hypomania and should always be implemented concurrently with pharmacotherapy (Shah et al., 2017).
Acute Bipolar II Depression
- Quetiapine (Level 1 — the only first-line agent for BD-II depression per CANMAT/ISBD 2023): 300 mg per day; robust evidence across BD-I and BD-II depression randomized controlled trials (RCTs) (Keramatian et al., 2023).
- Lithium (Level 2): Target serum level of 0.8 to 1.2 mEq/L. Evidence from BD-II-specific trials exists, though uncertainty remains about the magnitude of effect (Lundin et al., 2025; Keramatian et al., 2023).
- Lamotrigine (Level 2): Effective as an add-on treatment for depressive episodes. A 2024 systematic review and meta-analysis confirmed that adjunctive lamotrigine significantly reduced depressive symptomatology versus placebo (SMD -0.30; 95% CI: -0.51 to -0.10) and reduced the risk of depressive recurrence (RR: 0.78; 95% CI: 0.63 to 0.98) in bipolar depression (Haenen et al., 2024).
- Adjunctive antidepressants (Level 2 — restricted use): Sertraline or venlafaxine may be added to patients with pure (non-mixed) BD-II depression who are already receiving mood stabilizer coverage. Antidepressant monotherapy remains contraindicated (Keramatian et al., 2023; Shah et al., 2017).
- Lumateperone 42 mg per day (Level 2): FDA-approved for BD-I and BD-II depression. Included in the 2023 CANMAT/ISBD update as a second-line option given limited BD-II-specific data (Keramatian et al., 2023).
Maintenance Pharmacotherapy
- Lithium (Level 2): The preferred long-term agent for BD-II given its anti-suicidal properties and evidence for reducing both depressive and hypomanic recurrences. BD-II-specific evidence is of very low certainty (Lundin et al., 2025).
- Lamotrigine (Level 2): Preferred for maintenance when depression predominates the clinical course; superior for prevention of depressive recurrence (Haenen et al., 2024; Keramatian et al., 2023).
- Quetiapine (Level 2): Effective for maintenance with demonstrated reduction in both poles of illness.
- Combination therapy (lithium or valproate combined with lamotrigine or quetiapine): Considered when monotherapy proves insufficient.
Second-Line Pharmacotherapy
- Acute BD-II depression: Electroconvulsive therapy (ECT), particularly for severe, treatment-refractory depression or suicidal crisis, as well as the olanzapine-fluoxetine combination (OFC) and adjunctive lurasidone.
- Acute hypomania: Carbamazepine and divalproex.
- Maintenance: Valproate, carbamazepine, and aripiprazole (limited BD-II-specific evidence).
Third-Line and Adjunctive Options
- Clozapine: Considered for treatment-refractory BD-II with recurrent suicidal ideation. Requires mandatory hematological monitoring under a Risk Evaluation and Mitigation Strategy (REMS).
- Ketamine and Esketamine: Emerging evidence supports use in acute suicidal crises in bipolar depression. Not yet at guideline level, but clinically relevant for acute inpatient management (Berk et al., 2025).
- Pramipexole: A dopamine agonist with preliminary Level 2 evidence as adjunctive therapy for BD-II depression (Keramatian et al., 2023).
- Benzodiazepines (short-term only): Lorazepam or clonazepam for acute agitation or sleep dysregulation during hypomanic episodes. Not indicated for long-term use.
Important note: Antidepressant monotherapy, high-dose stimulants, and unaugmented monoamine oxidase inhibitors (MAOIs) are not recommended in BD-II given the risk of mood switch, cycling acceleration, and treatment-emergent mixed features (Keramatian et al., 2023; Shah et al., 2017).
Psychotherapy
Individual Therapy
Two individual psychotherapies hold the strongest evidence for BD-II and should be offered in combination with pharmacotherapy wherever possible.
Interpersonal and Social Rhythm Therapy (IPSRT): IPSRT holds the strongest BD-II-specific evidence of any psychotherapy. One small and one larger RCT support IPSRT as monotherapy for acute BD-II depression, and a systematic review confirms its efficacy for symptom reduction and relapse prevention in bipolar disorder (Berk et al., 2025). Its mechanism involves stabilizing social rhythms and sleep-wake cycles to reduce circadian disruption — the core biological vulnerability in BD-II.
Cognitive Behavioural Therapy (CBT): Evidence supports adjunctive CBT for BD-II depression and relapse prevention. A meta-analysis confirmed that CBT significantly reduced depressive symptoms compared to treatment as usual (SMD: -0.32; 95% CI: -0.64 to -0.01), with equal efficacy demonstrated for both BD-I and BD-II (Miklowitz et al., 2021; Nakagawa et al., 2022).
Individual Psychoeducation: Structured psychoeducation improves illness insight, medication adherence, recognition of prodromal symptoms, and reduces relapse rates. It is particularly important in BD-II given the common lack of insight into hypomanic states (Levrat et al., 2024).
Group Therapy
Group Psychoeducation: A 2021 network meta-analysis found that manualized psychotherapy combined with pharmacotherapy reduced illness recurrence compared to treatment as usual (OR: 0.56; 95% CI: 0.43 to 0.74). Family or conjoint therapy obtained the highest surface under the cumulative ranking curve (SUCRA 95%) for recurrence prevention (Miklowitz et al., 2021).
Family-Focused Therapy (FFT): FFT consists of three structured phases: psychoeducation, communication enhancement training, and problem-solving skills. It demonstrated equal efficacy to CBT and IPSRT for BD-I and BD-II depression in the STEP-BD trial, and is particularly beneficial when high expressed emotion or significant family dysfunction is present (Miklowitz et al., 2021; Levrat et al., 2024).
Complementary and Interprofessional Care
Nursing
Psychiatric nurses play a central role in BD-II management. Core functions include medication education and adherence monitoring, delivery of structured psychoeducation, MSE assessment at each encounter with vigilance for subtle hypomanic shifts, safety assessment and safety planning for suicidal ideation, and crisis intervention. Given that BD-II patients frequently present between episodes with residual depressive symptoms, nurses are often the first clinicians to detect early relapse signs. Collaborative care models with nurse care managers improve outcomes in bipolar disorder (Keramatian et al., 2023). Nurses should be familiar with the BD-II-specific risk of antidepressant-induced destabilization and counsel patients accordingly.
Nutrition
Metabolic comorbidities, including obesity, dyslipidemia, and type 2 diabetes are prevalent in BD-II, compounded by the weight-promoting effects of quetiapine, lithium, and valproate. Registered dietitian involvement is recommended for metabolic monitoring, nutritional counselling, and weight management support. Omega-3 fatty acids, particularly EPA-dominant formulations, have preliminary evidence as mood-stabilizing adjuncts in bipolar depression. Caffeine restriction and alcohol avoidance are clinically relevant behavioral recommendations (Keramatian et al., 2023; Berk et al., 2025). Nutritional screening using standardized tools such as Body Mass Index (BMI) monitoring should be routine practice.
Physiotherapy
Structured aerobic exercise is associated with reduced depressive symptoms, improved sleep quality, and enhanced cognitive function in bipolar disorder. A minimum of 150 minutes of moderate-intensity aerobic activity per week is recommended as an adjunct to standard treatment. Physiotherapists contribute by designing individualized, safe exercise programs, particularly for patients with metabolic syndrome, obesity, or cardiovascular comorbidities, all of which are elevated in BD-II. Exercise also serves as a non-pharmacological strategy for circadian rhythm stabilization, which is of particular therapeutic relevance in BD-II (Berk et al., 2025).
Social Work
Social workers address the psychosocial and structural determinants of health that disproportionately affect patients with BD-II: housing instability, financial strain, employment disruption, legal involvement, and social isolation. Core clinical functions include safety planning, crisis intervention, psychiatric advance directive development, system navigation, and connection to disability benefits and community mental health supports. Given the frequent misdiagnosis of BD-II as depression or personality disorder, social workers also play an important role in advocacy, stigma reduction, and ensuring continuity of care across service settings.
Occupational Therapy
Occupational therapists address the often substantial but underrecognized functional impairment in BD-II. Even during euthymia, cognitive deficits in attention, processing speed, and executive function reduce occupational performance. Core occupational therapy interventions include neurocognitive rehabilitation, activity scheduling, energy conservation, vocational rehabilitation, sleep hygiene structuring, and return-to-work planning. The regulation of daily routines, a cornerstone of occupational therapy directly supports the social rhythm stability that is central to BD-II relapse prevention.
Evaluation of Bipolar II Disorder
Prognosis
BD-II is a chronic, recurrent condition with a predominantly depressive course. Prognosis is frequently more complex than initially anticipated. Even with guideline-recommended treatment, 20 to 30% of patients have a poor long-term prognosis, and subsyndromal depressive symptoms persist between episodes in the majority of individuals (Berk et al., 2025; Hu et al., 2025).
Key prognostic indicators include the following. Negative prognostic factors include rapid cycling, mixed features, comorbid anxiety or substance use disorders, a high number of prior episodes, early illness onset, poor medication adherence, and lack of social support. Positive prognostic factors include sustained pharmacotherapy adherence, engagement with IPSRT or other structured psychotherapy, stable daily routines, absence of comorbid substance use, and strong interpersonal support networks.
Suicide risk must be explicitly reassessed at every clinical encounter, as BD-II carries equivalent completed suicide rates to BD-I (pooled OR = 1.00; Dev et al., 2024). Lithium’s anti-suicidal properties make it a preferred agent in BD-II patients with a history of suicidal behaviour (Shah et al., 2017).
Follow-Up
After a hypomanic or depressive episode, follow-up should occur within one to two weeks of discharge or treatment initiation. Comprehensive monitoring should include the following components.
- Serum lithium levels every three to six months during stable periods; thyroid function (TSH) and renal function (eGFR) every six to twelve months for lithium-treated patients.
- Metabolic monitoring, including fasting glucose, lipid profile, weight, waist circumference, and blood pressure — every three months initially, then annually, for patients on second-generation antipsychotics.
- Lamotrigine: no required serum levels, but vigilant monitoring for dermatological reactions, especially during titration.
- Mood symptom monitoring with the MADRS or PHQ-9 for depression and the HCL-32 or YMRS for hypomanic symptoms at each visit.
- Life chart review: sleep patterns, social rhythm regularity, stressors, substance use, and medication adherence.
- Suicide risk assessment at every clinical encounter, given the persistently elevated risk in BD-II.
- Review of advance directives and relapse prevention plans at least annually and after each episode.
CANMAT/ISBD 2023 guidelines recommend maintenance pharmacotherapy for a minimum of two years after a first episode of BD-II, and indefinitely in patients with multiple episodes, significant suicidal history, or marked functional impairment (Keramatian et al., 2023). Shah et al. (2017) similarly advocate for long-term maintenance to prevent the progressive cognitive and functional deterioration associated with recurrent mood episodes.
Referral
Clinicians should consider referral to or consultation with the following specialists.
- Psychiatrist: Required for diagnostic confirmation (especially when BD-II versus BPD or MDD is uncertain), complex pharmacotherapy initiation, treatment-resistant depression, and acute hospitalization decisions.
- Neurologist: When a medical or neurological etiology is suspected, such as epilepsy, autoimmune encephalitis, CNS lesion, or hypothyroidism presenting as depression.
- Addiction medicine or concurrent disorders program: Comorbid substance use disorder affects approximately 30.7% of bipolar patients and significantly worsens prognosis (Léda-Rêgo et al., 2024).
- ECT service: For pharmacotherapy-resistant BD-II depression, severe suicidal ideation, or depressive episodes with catatonic or psychotic features.
- Perinatal psychiatry: Mandatory consultation during pregnancy, given the teratogenic risk of valproate (absolutely contraindicated) and lithium. Postpartum monitoring is essential given the elevated BD-II relapse risk in the postpartum period.
- Neuropsychology: For formal neurocognitive assessment when euthymic cognitive impairment affects occupational function, is unexplained, or requires documentation for disability or accommodation purposes.
Red Flags
The following situations require urgent or emergent clinical response.
- Active suicidal ideation with plan, intent, or a recent attempt: requires immediate safety assessment, evidence-based safety planning, and urgent risk-stratified intervention. Clinicians must not minimize suicide risk in BD-II based on diagnosis alone (Dev et al., 2024).
- Escalation of a hypomanic episode to full mania, including new or worsening psychosis, a need for hospitalization, or complete loss of insight: indicates possible BD-I and requires urgent psychiatric assessment and BD-I-level treatment.
- Lithium toxicity at serum levels above 1.5 mEq/L: symptoms include coarse tremor, ataxia, confusion, vomiting, and diarrhea. This is a medical emergency requiring immediate cessation of lithium, intravenous hydration, and hospitalization.
- Antidepressant-induced mood switch: new or escalating hypomanic or manic symptoms, mixed features, or rapidly cycling mood following antidepressant initiation or dose increase. Requires reassessment of diagnosis and treatment plan.
- Lamotrigine-associated Stevens-Johnson syndrome or toxic epidermal necrolysis: any new rash during lamotrigine titration requires immediate medication cessation and urgent dermatological assessment.
- Valproate-induced hyperammonemic encephalopathy: confusion, vomiting, or lethargy at any dose requires immediate investigation and medication cessation.
- Pregnancy in a patient on valproate: an absolute contraindication requiring urgent perinatal psychiatry and obstetric consultation.
- Emerging psychosis in a patient with a BD-II diagnosis: requires re-evaluation of the diagnosis of BD-I, schizoaffective disorder, or an organic etiology — and urgent psychiatric consultation.
Resources
- Canadian Network for Mood and Anxiety Treatments (CANMAT)
- International Society for Bipolar Disorders (ISBD)
- Depression and Bipolar Support Alliance (DBSA)
- National Alliance on Mental Illness (NAMI)
- World Health Organization: Bipolar Disorder Fact Sheet
- Indian Psychiatric Society: Clinical Practice Guidelines
References
- American Psychiatric Association. (2022). Diagnostic and statistical manual of mental disorders (5th ed., text rev.). https://doi.org/10.1176/appi.books.9780890425787
- Berk, M., Corrales, A., Trisno, R., Dodd, S., Yatham, L. N., Vieta, E., McIntyre, R. S., Suppes, T., & Agustini, B. (2025). Bipolar II disorder: A state-of-the-art review. World Psychiatry, 24(2), 175–189. https://doi.org/10.1002/wps.21300
- Dev, D. A., Le, G. H., Kwan, A. T. H., Wong, S., Arulmozhi, A., Ceban, F., Teopiz, K. M., Meshkat, S., Rosenblat, J. D., Guillen-Burgos, H. F., Rhee, T. G., Ho, R. C., Cao, B., d’Andrea, G., Sundberg, I., & McIntyre, R. S. (2024). Comparing suicide completion rates in bipolar I versus bipolar II disorder: A systematic review and meta-analysis. Journal of Affective Disorders, 361, 480–488. https://doi.org/10.1016/j.jad.2024.06.045
- Fico, G., De Prisco, M., Oliva, V., Vieta, E., & Lejoyeux, M. (2025). Bipolar disorders: An update on critical aspects. The Lancet Regional Health – Europe, 49, 101135. https://doi.org/10.1016/j.lanepe.2024.101135
- Haenen, N., Kamperman, A. M., Prodan, A., Nolen, W. A., Boks, M. P., & Wesseloo, R. (2024). The efficacy of lamotrigine in bipolar disorder: A systematic review and meta-analysis. Bipolar Disorders, 26(5), 431–441. https://doi.org/10.1111/bdi.13452
- Hu, Y., Xie, J., Liu, M., et al. (2025). Global, regional, and national burden of bipolar disorder, 1990–2021: Analysis of data from the Global Burden of Disease Study 2021. Journal of Affective Disorders. Advance online publication. https://doi.org/10.1016/j.jad.2025.10.035
- Keramatian, K., Chithra, N. K., & Yatham, L. N. (2023). The CANMAT and ISBD guidelines for the treatment of bipolar disorder: Summary and a 2023 update of evidence. Focus, 21(4), 394–416. https://doi.org/10.1176/appi.focus.20230009
- Léda-Rêgo, G., Studart-Bottó, P., & Abbade, P. (2024). Lifetime prevalence of psychiatric comorbidities in patients with bipolar disorder: A systematic review and meta-analysis. Journal of Affective Disorders, 355, 123–132. https://doi.org/10.1016/j.jad.2024.02.073
- Levrat, V., Favre, S., & Richard-Lepouriel, H. (2024). Current practices of psychoeducation interventions with persons with bipolar disorders: A literature review. Frontiers in Psychiatry, 14, 1320654. https://doi.org/10.3389/fpsyt.2023.1320654
- Lundin, J., Ekström, E., & Vieta, E. (2025). Lithium and lamotrigine for the treatment of bipolar II disorder – A systematic review and meta-analysis of randomized trials. Journal of Affective Disorders. Advance online publication. https://doi.org/10.1016/j.jad.2025.04.026
- Miklowitz, D. J., Efthimiou, O., Furukawa, T. A., Scott, J., McLaren, R., Geddes, J. R., & Cipriani, A. (2021). Adjunctive psychotherapy for bipolar disorder: A systematic review and component network meta-analysis. JAMA Psychiatry, 78(2), 141–150. https://doi.org/10.1001/jamapsychiatry.2020.2993
- Mullins, N., Forstner, A. J., O’Connell, K. S., et al. (2021). Genome-wide association study of more than 40,000 bipolar disorder cases provides new insights into the underlying biology. Nature Genetics, 53(6), 817–829. https://doi.org/10.1038/s41588-021-00857-4
- Nakagawa, A., Sado, M., Mitsuda, D., Fujisawa, D., Kikuchi, T., Abe, T., & Mimura, M. (2022). Effectiveness of cognitive behavioural therapy augmentation of pharmacotherapy in reducing depressive symptoms in patients with bipolar I or II disorder: Meta-analysis of randomized controlled trials. Frontiers in Psychiatry, 12, 781583. https://doi.org/10.3389/fpsyt.2021.781583
- Radua, J., De Prisco, M., Oliva, V., Fico, G., Vieta, E., & Fusar-Poli, P. (2024). Impact of air pollution and climate change on mental health outcomes: An umbrella review of global evidence. World Psychiatry, 23(2), 244–256. https://doi.org/10.1002/wps.21219
- Shah, N., Grover, S., & Rao, G. P. (2017). Clinical practice guidelines for management of bipolar disorder. Indian Journal of Psychiatry, 59(Suppl 1), S51–S66. https://doi.org/10.4103/0019-5545.196974